How PL3D Works?
- Precise Access - A fine 21G needle is guided into the affected disc under imaging.
- Laser Activation - A micro-laser fiber delivers controlled energy to vaporize a tiny portion of disc tissue.
- Pressure Relief - Intradiscal pressure drops immediately, relieving nerve compression.
- Smoke Evacuation (Patented System) - Gas produced is immediately removed by a built-in smoke evacuation system (Y-connector), minimizing postoperative muscle spasm.Safety Assured - Temperature inside the disc is kept below 50 °C to protect nerves.
- Controlled Thermal Safety
· Low heat spread.
· 980 nm wavelength provides high water absorption leads efficient ablation with minimal collateral heating (no discitis).
Because of its low operating temperature (<50 °C), PL3D avoids thermal injury, scarring, or spinal instability. Patients typically walk out the same day and return to work within 5–7 days.
|
Minimally invasive |
21G needle access into the disc — no incision, no bone removal, no muscle damage, no peridural scarring. Allows disc decompression without affecting spinal stability. |
|
Short recovery |
Outpatient procedure; patients usually return to normal routine quickly with significantly less morbidity compared to open surgery. Suitable even for high-risk cardiac/renal/hepatic patients because only local anaesthesia is used |
|
Safety built-in |
Real-time temperature control < 50 °C, fiber verification, and laser output testing |
|
Active suction |
Patented Y-connector removes intradiscal gas created during vaporization, preventing pressure build-up and reducing postoperative muscle spasm. |
|
980 nm wavelength |
980 nm absorption prevents discitis; external power meter confirms true fiber output; sterile fiber testing system ensures safe operation before insertion. 10× higher absorption vs 810 nm and 5× vs 1064 nm, allowing effective vaporization with lower energy and minimal collateral heat leads safer for nerves and surrounding tissues. |
|
Precision laser control |
Operator-defined pulse settings (power, exposure time, joules) based on disc pathology for controlled, targeted decompression without damaging healthy tissue. |

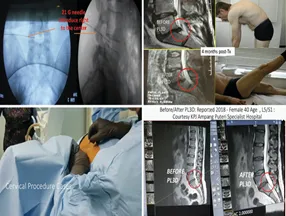
Clinical Safety and Efficacy
- FDA-approved since 1991 and performed in over 80,000 cases worldwide
- Success rate: 89 ≈ 91 %
- Complication rate: < 1 %
- Uses 10–15 W laser power and maintains temperature below 50 °C for maximum safety
- Features a patented smoke-evacuation system to control pressure and prevent burns
- Patients typically walk within hours and return to normal activity in 5–7 days